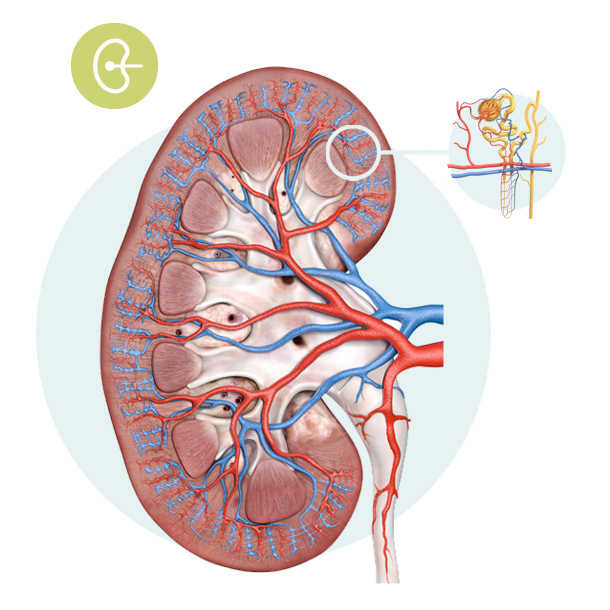
Kidney
The kidney is the organ that filters the blood. Blood to be filtered comes via the renal artery (in red), blood filtered leaves in the renal vein (in blue), and the waste products are eliminated in the urine.
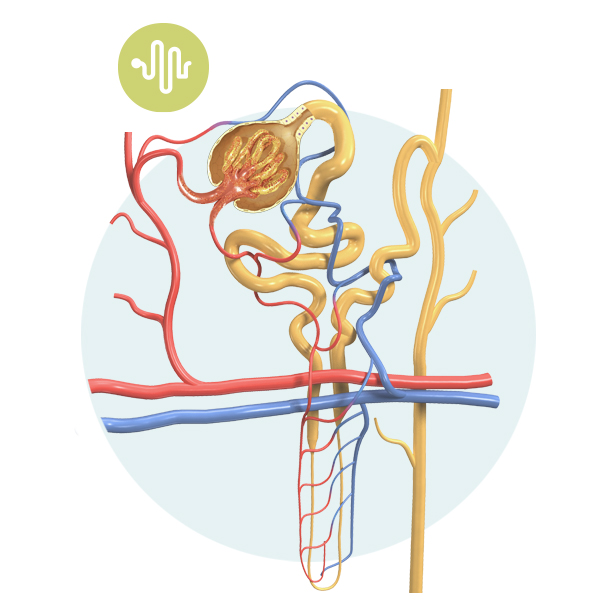
Nephron
Each kidney is divided into individual functional units called nephrons. If the kidney is like a factory, the nephrons are the individual workers, and there are close to one million nephrons per kidney.
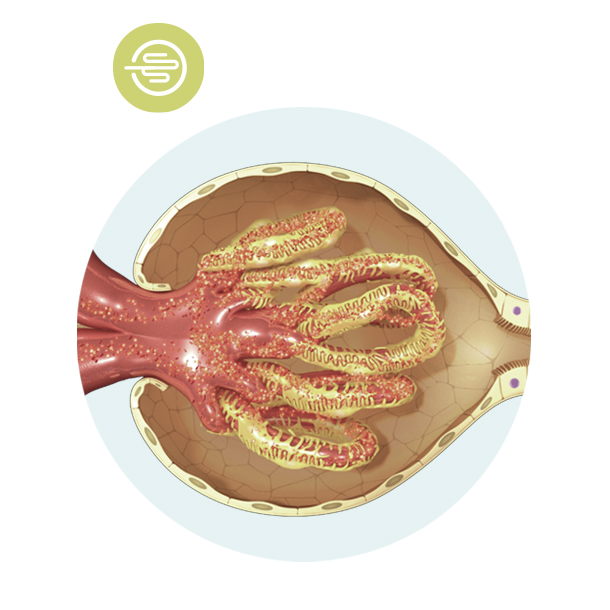
Glomerulus
The glomerulus is a key element of the nephron where important filtration mechanisms happen. The glomerulus is like the hand of the worker, deciding what is filtered out and what is kept in the blood.
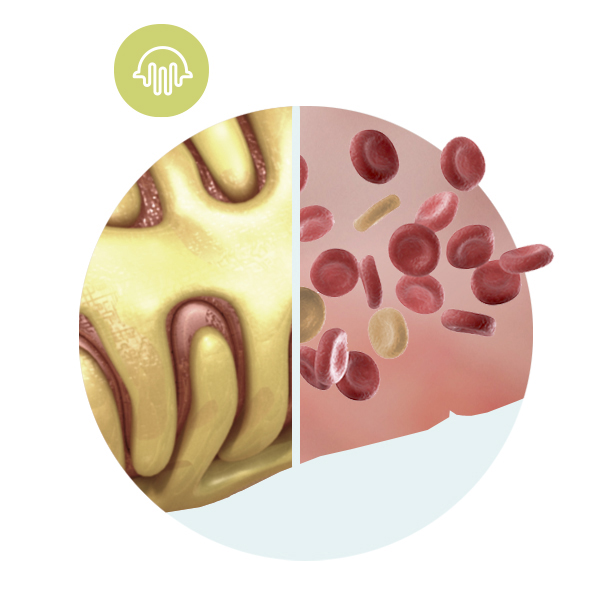
Podocyte
The podocyte is the key cell type responsible for the filtration of the blood at a cellular level. The podocytes are like the fingers of a hand, they wrap around blood vessels like interlocking fingers.
PODOCYTES
The podocyte – the key to kidney health.
The podocyte is a highly specialised kidney cell type implicated in many kidney diseases. We are the first company to treat kidney diseases by directly targeting the podocyte with AAV gene therapy.
The kidneys perform an essential job filtering the blood. Podocytes make up one of the most critical components of that filtering apparatus. The podocytes have long cellular extensions called foot processes or pedicels, after which the cells are named.
The pedicels wrap around the blood vessels and leave little gaps called slits between them. Blood is filtered through these gaps, each known as a filtration slit or slit diaphragm (in other words, analogous to the spaces between the teeth of a zipper).
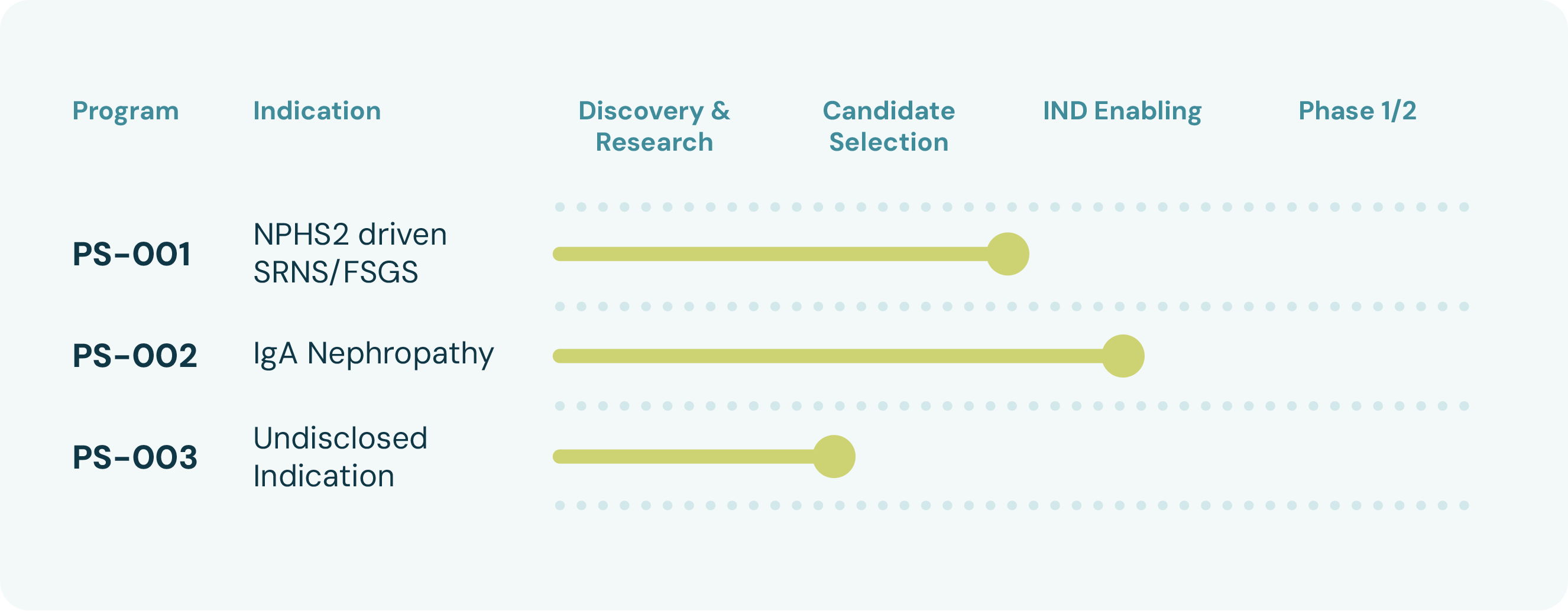
OUR STRONG PIPELINE
Our goal is to bring about a step change in gene therapy development.
Our growing pipeline has programmes in development targeting both rare monogenic diseases and more prevalent non-genetic diseases. Both types of diseases can lead to end-stage renal disease, with devastating consequences to patients. The lead program PS-002 is focused on complement driven glomerular diseases and will initially be developed for IgA nephropathy.
Our flexible platform allows us to leverage our capability to develop therapies for high unmet need glomerular diseases. Examples of such diseases are monogenic podocyte diseases (e.g. NPHS2 driven steroid resistant nephrotic syndrome and focal segmental glomerulosclerosis), glomerular basal membrane disorders and diabetic kidney disease.
PUBLICATIONS/PRESENTATIONS
Sharing our scientific results to advance innovation in the field.
We are committed to transparency and sharing our scientific results with the public to advance innovation in the field.
Recent publications and/or presentations from our team include:
Wen Y. Ding et al., Adeno-associated virus gene therapy prevents progression of kidney disease in genetic models of nephrotic syndrome. Sci. Transl. Med. 15, eabc8226 (2023). DOI:10.1126/scitranslmed.abc8226






